Explore the Architecture
Click the numbered hotspots on the left to examine
the molecular toolkit of a dendritic cell.
01
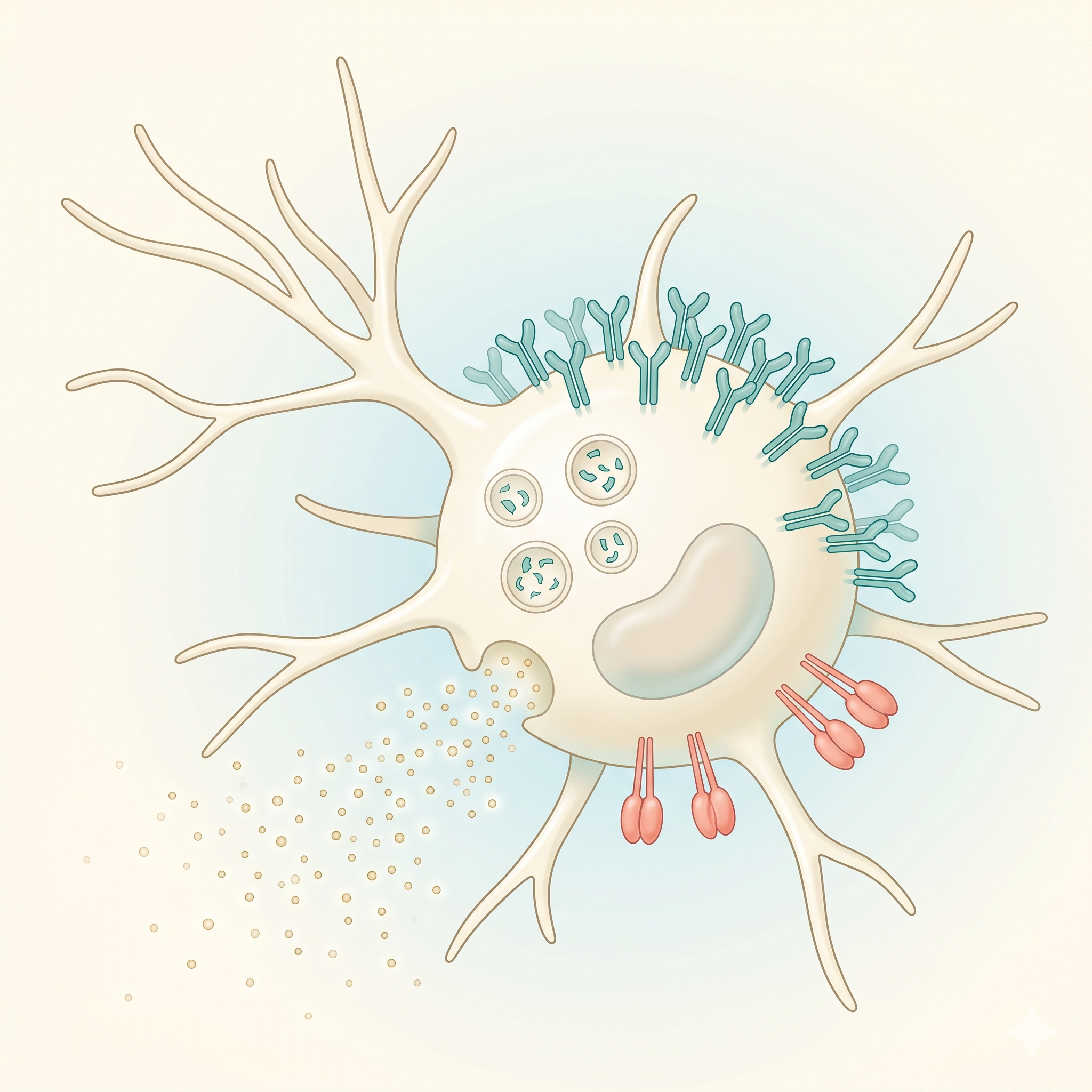
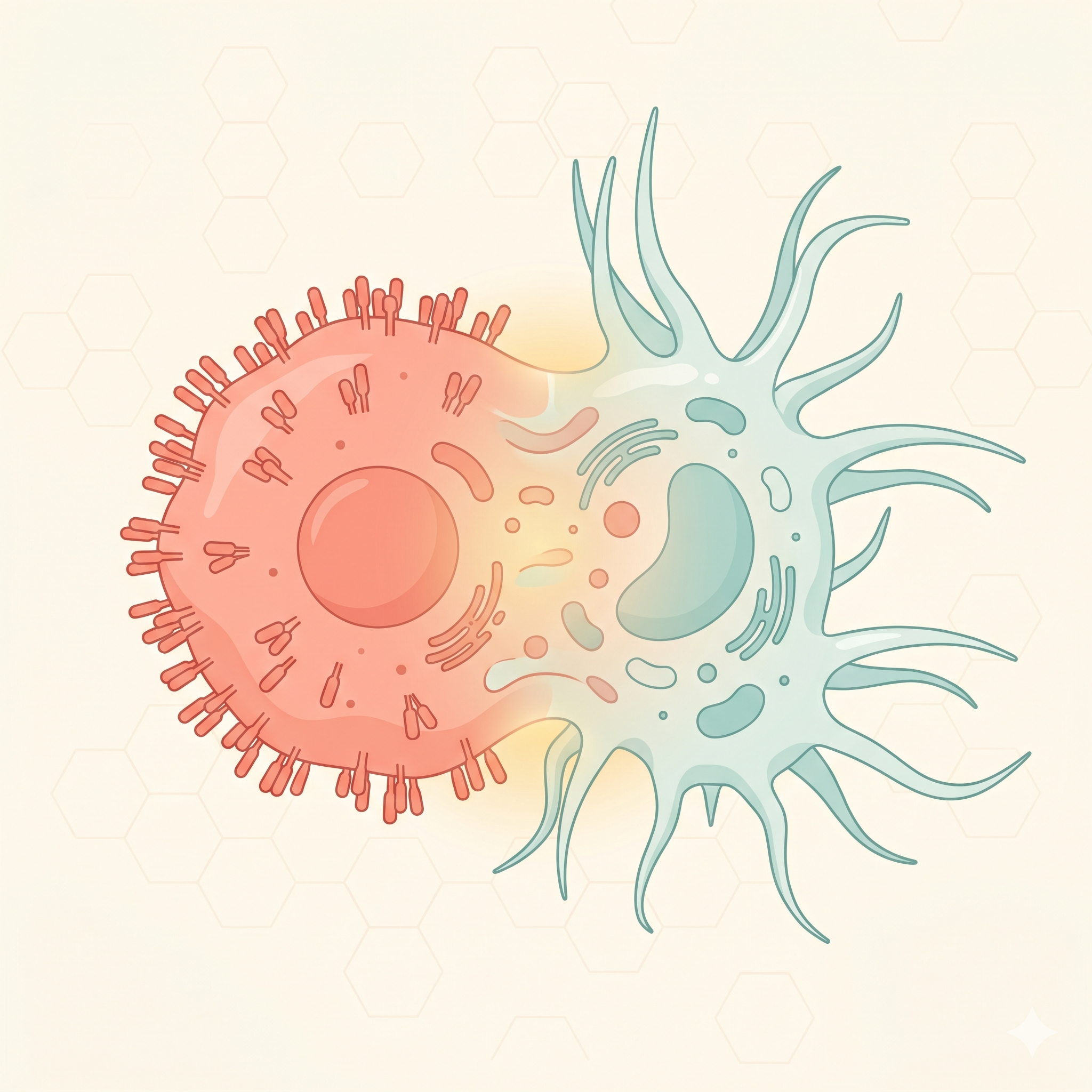
Dendrites
Star-shaped cytoplasmic projections
The long, branching cytoplasmic extensions that give the cell its
characteristic shape. They dramatically increase surface area, maximizing
antigen capture efficiency. A single DC can interact
with thousands of T cells simultaneously.
~10x
larger surface area than a spherical cell
02
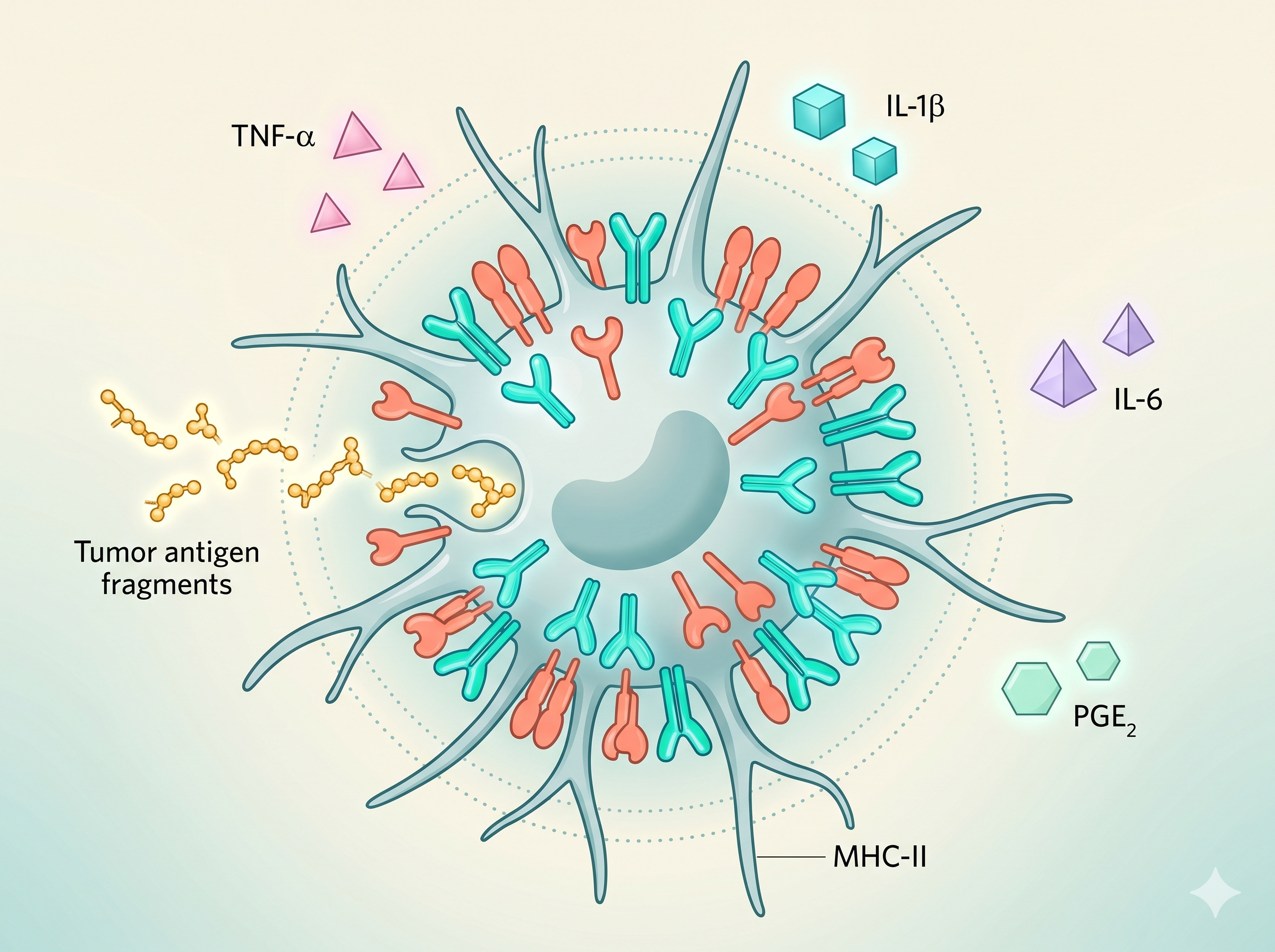
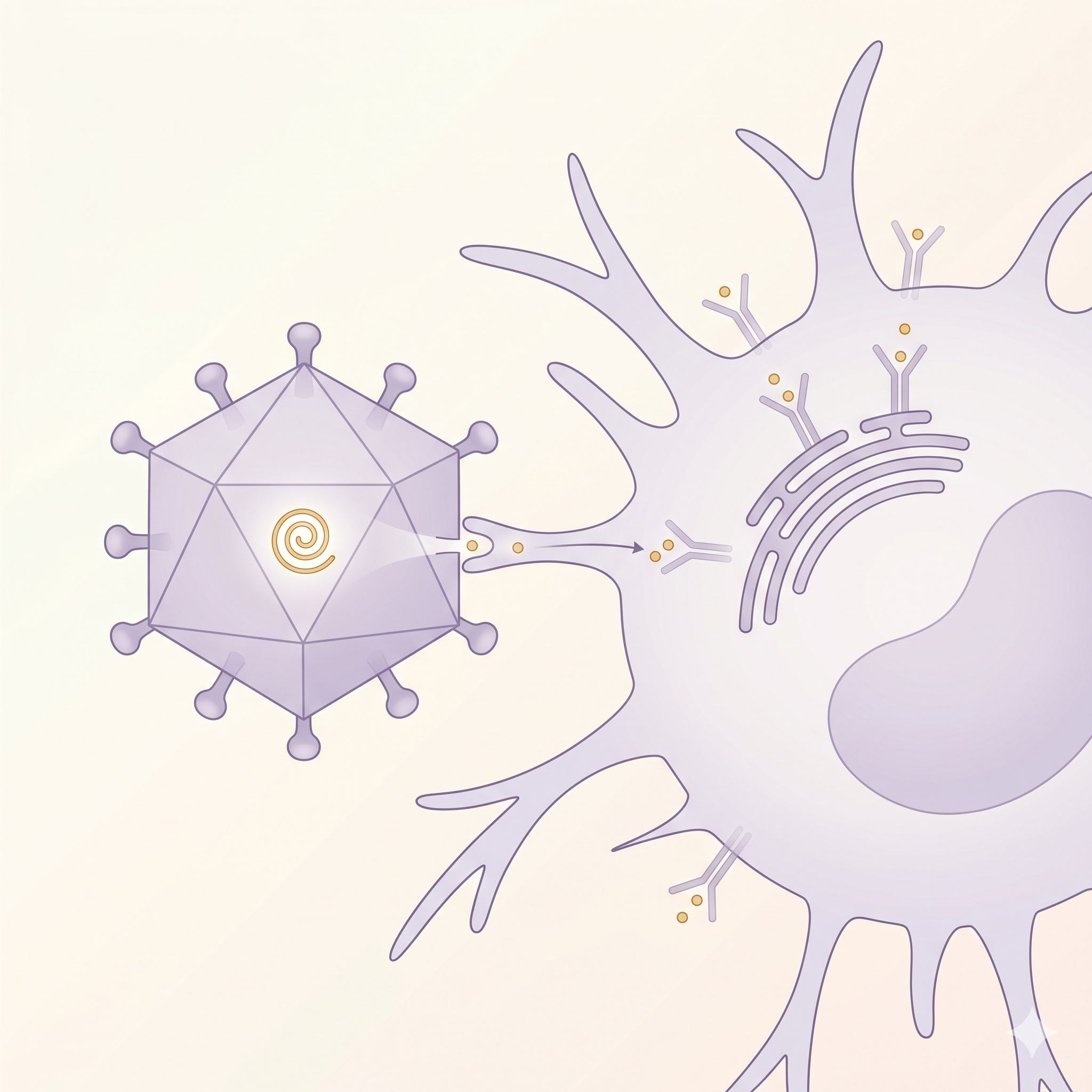
MHC-II Molecules
HLA-DR, HLA-DP, HLA-DQ
Major histocompatibility complex class II molecules that present
processed antigen peptides to CD4+ T cells. Surface expression
increases 10-100 fold upon DC maturation — a
hallmark of antigen presentation capacity.
10⁶+
surface MHC-II molecules per mature DC
03
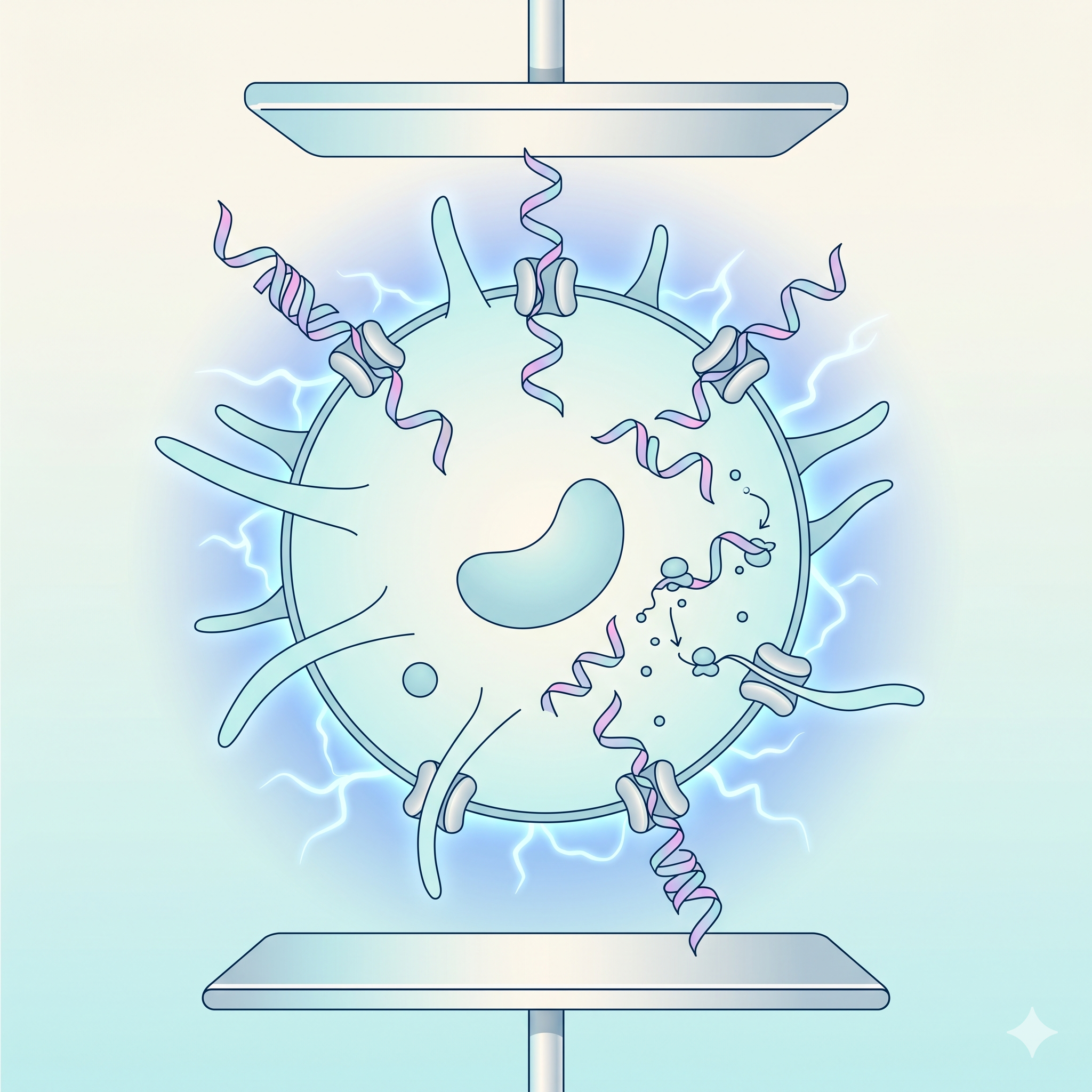
Costimulatory Molecules
CD80 (B7-1), CD86 (B7-2), CD40
The "second signal" molecules required for T cell
activation. Without them, T cells become anergic (unresponsive).
DCs' ability to upregulate these molecules at high levels is the
critical feature that distinguishes them from other antigen-
presenting cells.
CD28
the costimulator receptor partner on T cells
04
Cytokine Secretion
IL-12, IL-6, IFN-α, TNF-α
The DCs' "third signal" — cytokines secreted by DCs determine the
differentiation fate of T cells. IL-12 drives Th1
(anti-tumor) responses; IL-4 drives Th2 (humoral) responses. The
anti-tumor cytokine profile of mature DCs is the molecular basis
of cancer immunotherapy.
IL-12
strongest known Th1 / cytotoxic T cell inducer
05
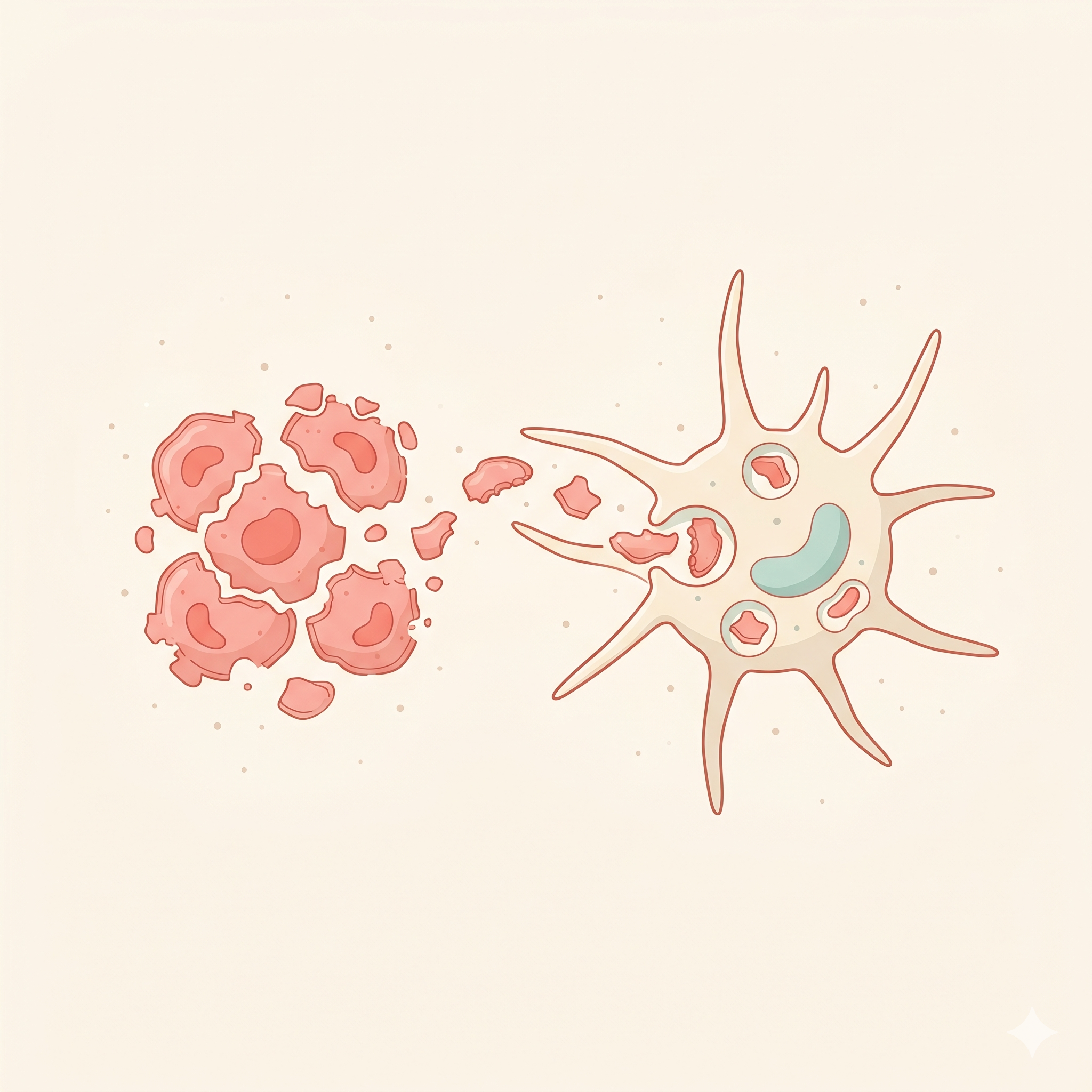
Phagocytic Vesicles
Endosome & phagolysosome
DCs capture extracellular antigens via macropinocytosis, phagocytosis,
and receptor-mediated endocytosis. Within these vesicles, antigens are
broken down into peptide fragments and loaded onto
MHC molecules. DCs' cross-presentation capability allows them
to display extracellular antigens on MHC-I to CD8+ T cells —
critical for anti-tumor immunity.
~24h
from antigen loading to mature presentation