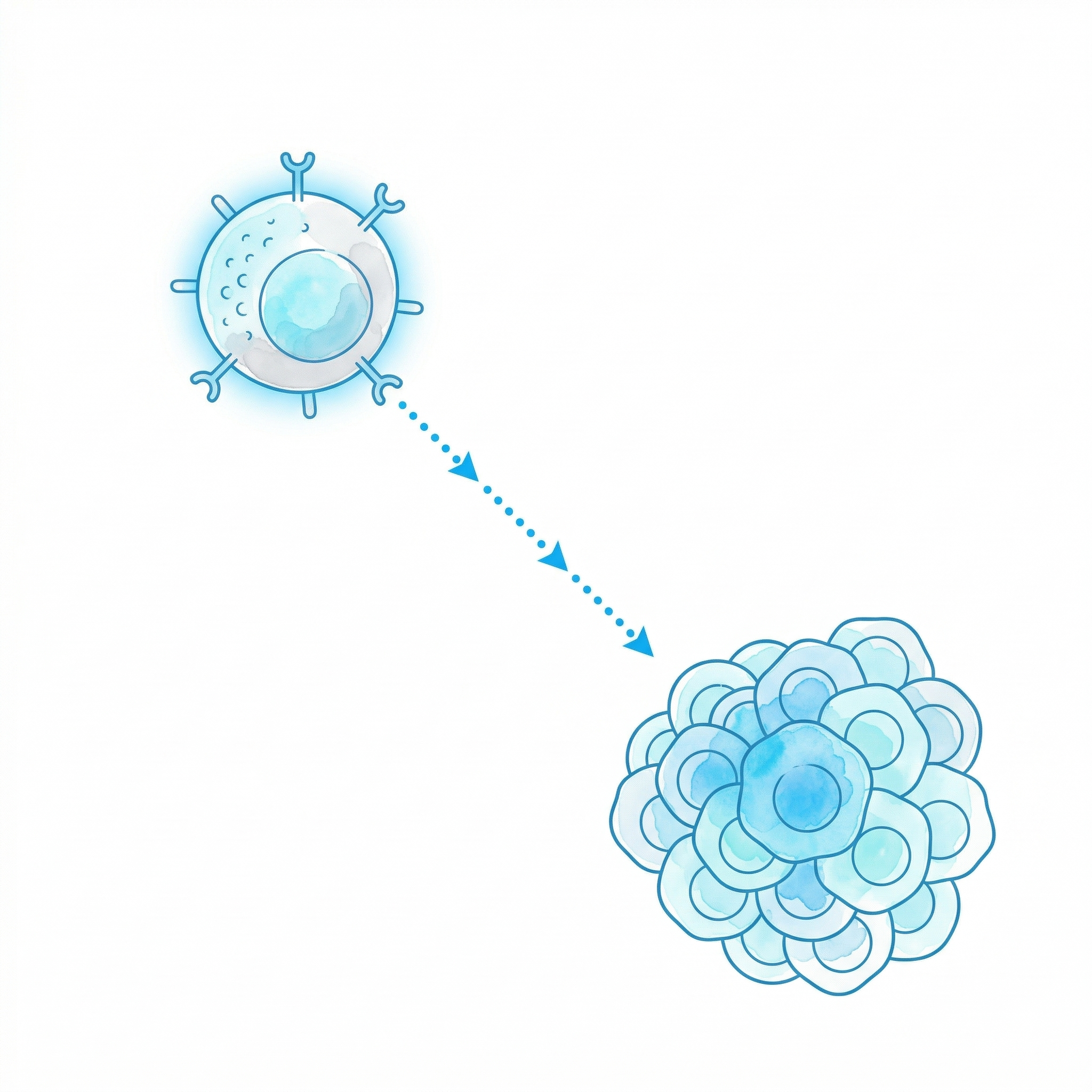
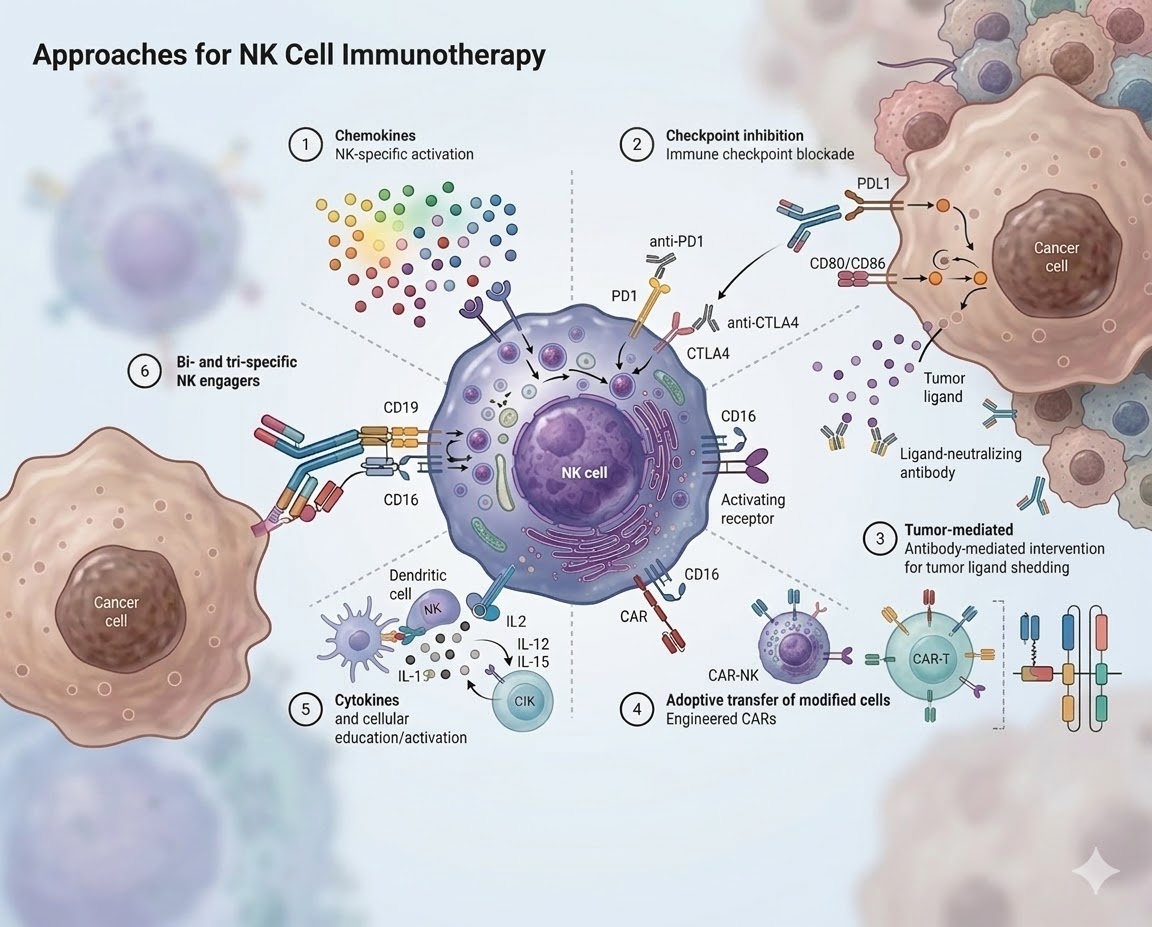
Chemokines
Specific recruitment and activation of NK cells into the tumor microenvironment. Targeted homing via CXCR3 and CX3CR1 receptors.
Cell Migration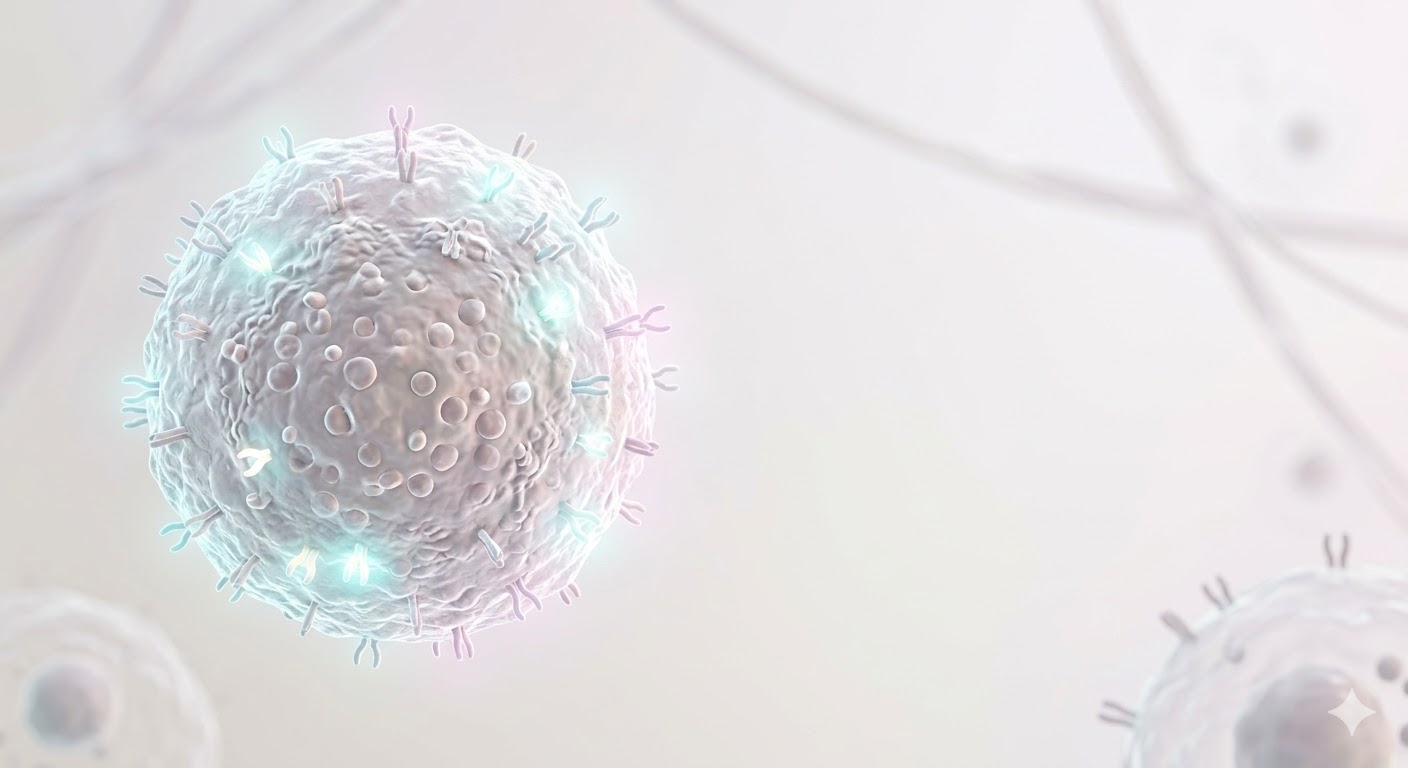
The elite warriors of the innate immune system that instantly recognize and destroy targets without prior sensitization.
Explore the Mechanism of Action ↓Natural Killer (NK) cells are the most potent effector cells of the innate immune system. They recognize and eliminate virus-infected and tumor cells without requiring prior activation or sensitization.
Click the numbered points on the left to examine different regions of the NK cell.
NKG2D, NKp30, NKp44, NKp46
Molecular sensors that recognize ligands on the surface of stressed, infected, or malignant cells. When these receptors are activated, the NK cell receives a "kill" signal.
KIR, NKG2A/CD94
These receptors recognize MHC class I molecules on the surface of healthy cells and send a "don't kill" signal to the NK cell. This balance protects normal cells — the "self-recognition" principle.
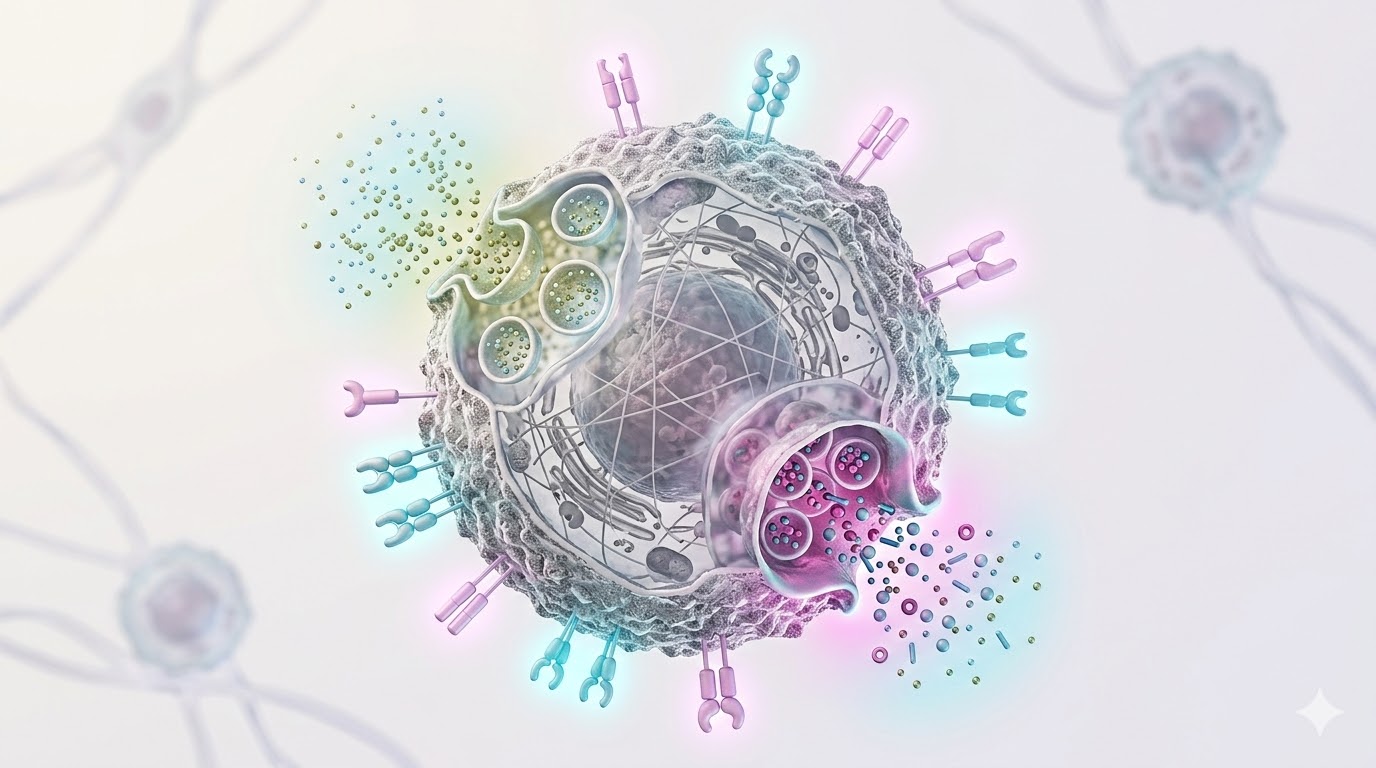
Perforin & Granzyme B
The NK cell's arsenal. Perforin punches holes in the target cell membrane, while Granzyme B enters through these pores and triggers programmed cell death (apoptosis). Like a serial killer, a single NK cell can eliminate multiple targets in succession.
IFN-γ, TNF-α, GM-CSF
NK cells do more than kill; they also secrete chemical messengers (cytokines) that recruit and activate other immune cells. IFN-γ in particular activates macrophages and strengthens the overall immune defense.
Lipid bilayer membrane
The fluid lipid membrane surrounding the NK cell carries hundreds of receptors and co-receptors on its surface. Membrane fluidity is critical for target adhesion (immunological synapse formation) and granule release.
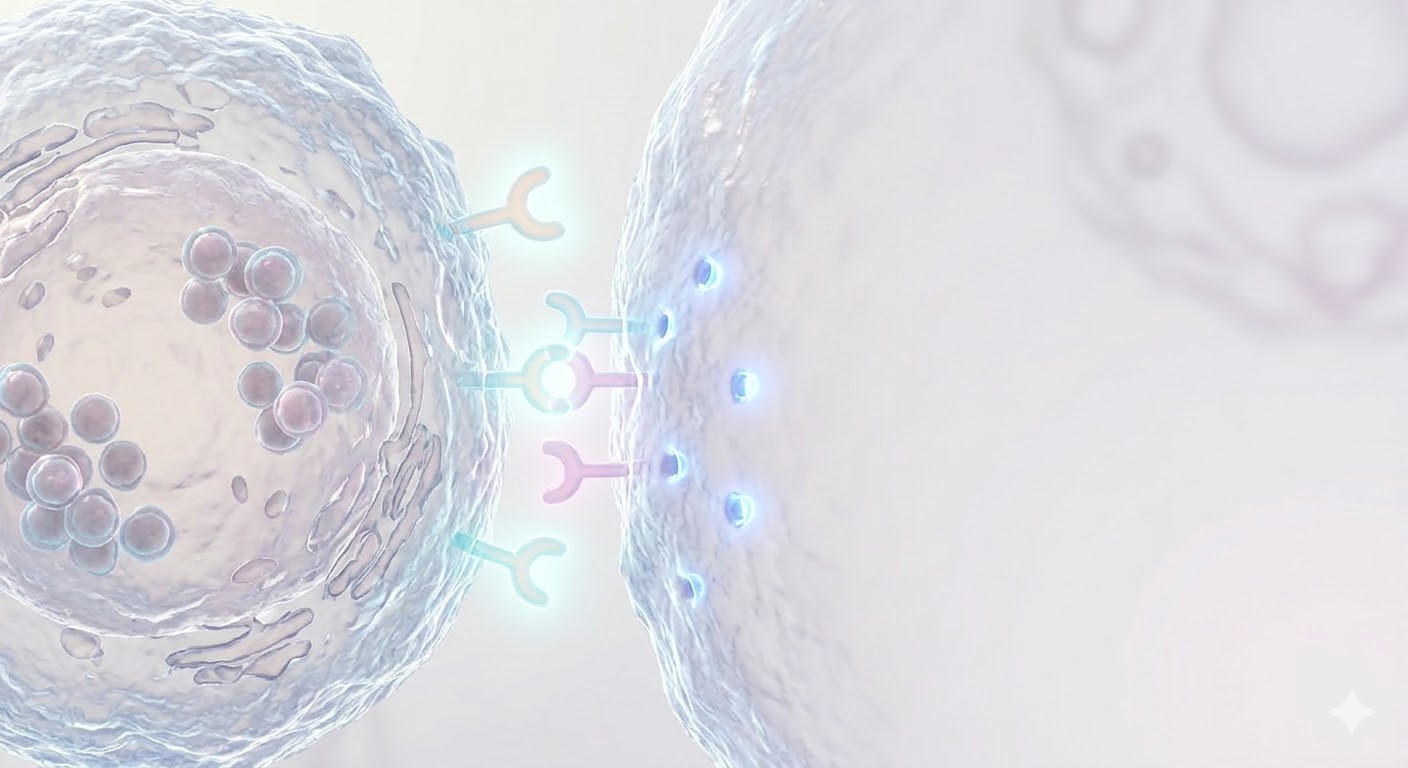
A precise three-phase destruction process that begins the moment an NK cell detects its target. Each step is a direct continuation of the previous one.
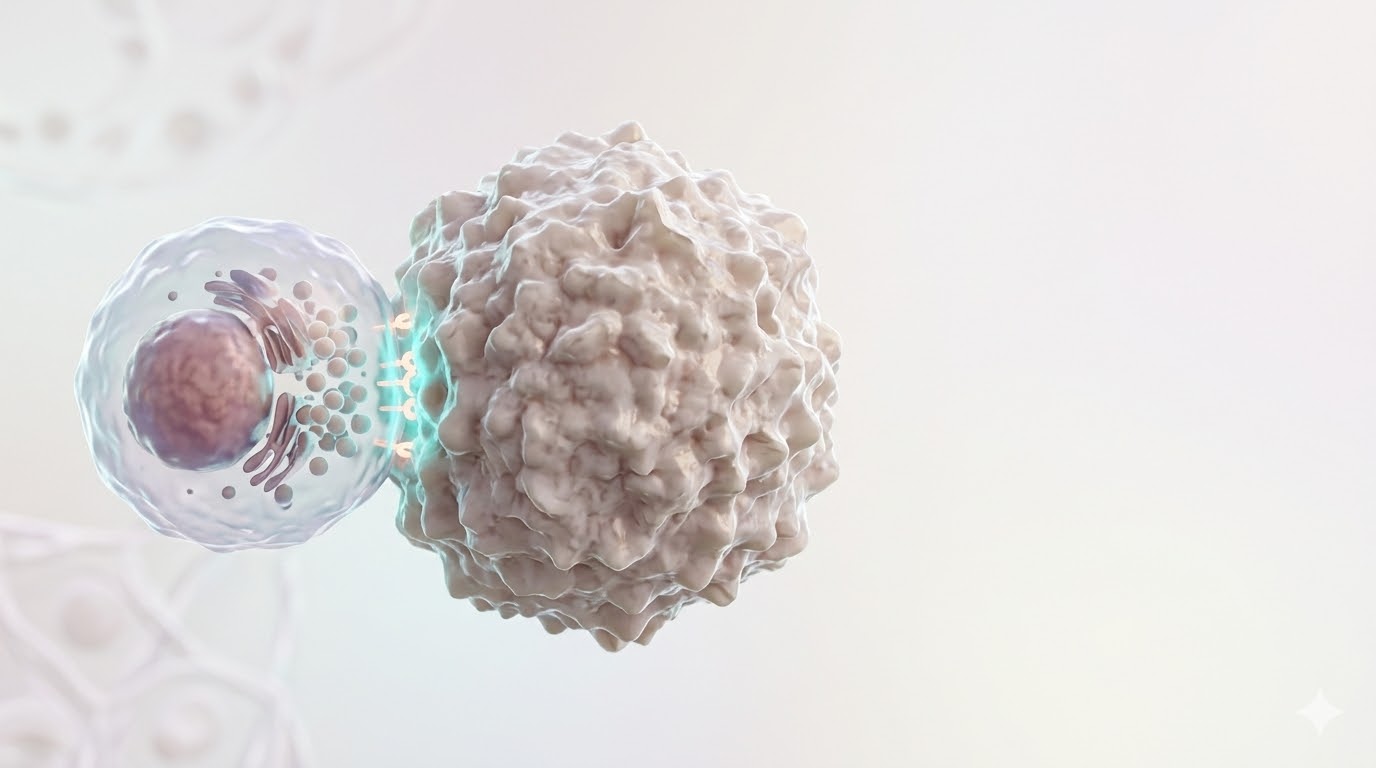
Locking onto the Target Cell
The NK cell recognizes stress ligands on the target cell surface via activating receptors (NKG2D, NKp30). A specialized contact zone known as the "immunological synapse" forms between the two cells. Actin filaments reorganize to create a tight adhesion ring — the target can no longer escape.
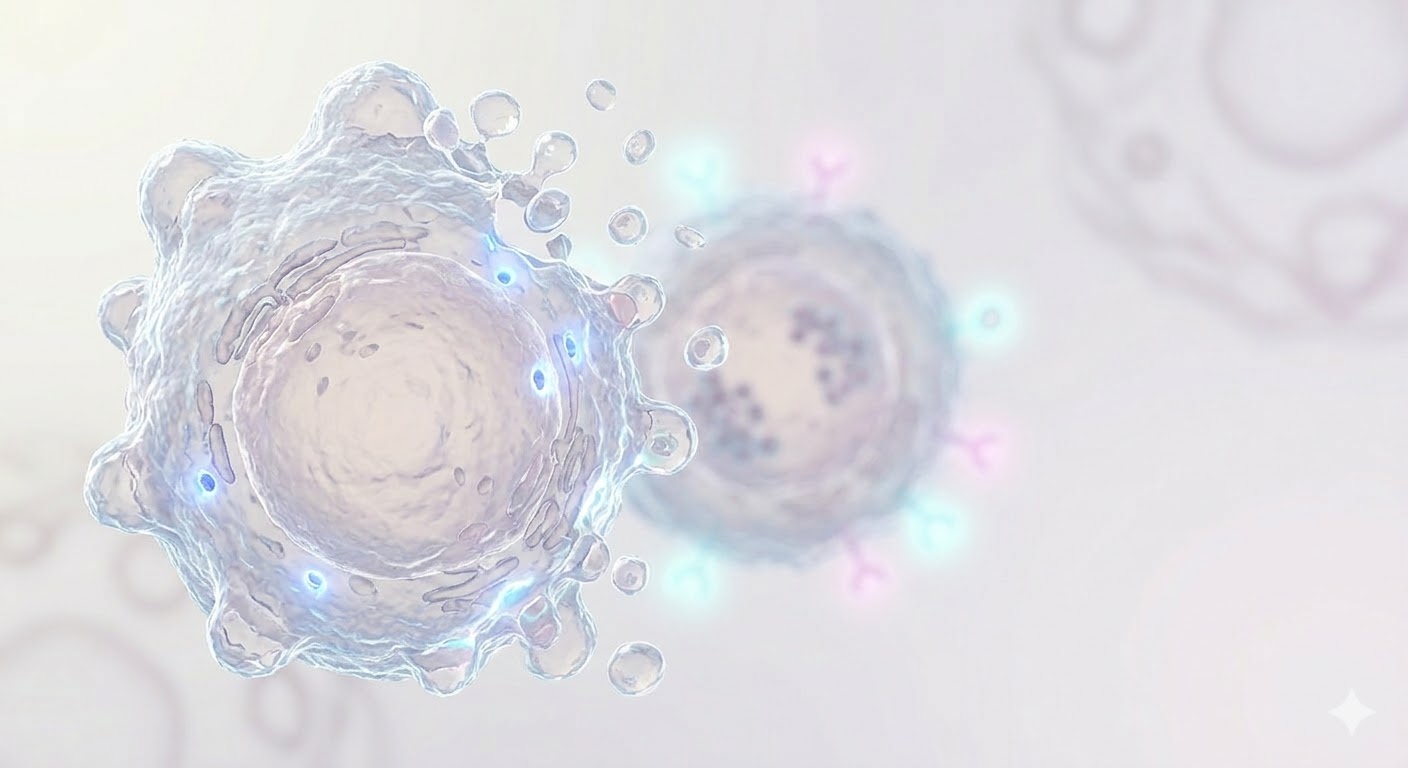
Armor-Piercing Strike
Once the synapse forms, the NK cell directs its lytic granules toward the synapse zone. Perforin molecules within the granules embed in the target cell membrane and create nanometer-scale pores. These pores serve as gateways for granzyme enzymes to enter the cell.
Programmed Cell Death
Granzyme B enzymes enter through the perforin pores and trigger the caspase cascade, initiating apoptosis (programmed cell death). DNA fragmentation and cell shrinkage follow. The NK cell detaches unharmed and, like a serial killer, moves on to the next target.
Data compiled from clinical trials worldwide demonstrate the power of NK cells in immunotherapy.
11 patients with advanced lymphoma and leukemia received NK cells engineered to recognize the CD19 protein on cancer cell surfaces (CAR-NK). The results:
• 8 of 11 patients (73%) showed tumor shrinkage (objective response)
• 7 of 11 patients (64%) achieved complete remission
Bottom line: Common CAR-T side effects — cytokine storm (excessive immune reaction), neurological toxicity, and tissue rejection (GvHD) — were not observed in any patient. Both effective and safe.
Liu et al., N Engl J Med, 2020; 382:545-553NK-cell-based cancer therapy is being tested worldwide. Per the U.S. registry of all clinical research, ClinicalTrials.gov:
• 420 NK trials registered by end of 2021
• ~47 new trials launched yearly since 2016
• Current count has surpassed 500
Bottom line: NK cell therapy is one of the fastest-growing research fields in oncology. Double-digit annual growth reflects both academic interest and commercial investment potential.
Myers & Miller, J Hematol Oncol, 2021; 14:183NK cell therapy is being studied across 20+ different cancer types:
• Blood cancers: acute/chronic leukemias (AML, ALL), lymphoma (NHL), multiple myeloma
• Solid tumors: lung, ovarian, pancreatic, brain (glioblastoma), melanoma, colorectal, breast
Distribution of CAR-NK studies: 54% blood cancers, 34% solid tumors, 11% autoimmune diseases.
Bottom line: Because NK cells aren't fooled by cancer's "identity hiding" trick (MHC loss), they reach a broad tumor spectrum — including tumors other immune cells can't.
Gong et al., Exp Hematol Oncol, 2024; 13:18High-resolution single-cell imaging measured the time between NK contact and tumor cell death:
• Within minutes of contact, the NK cell secretes a pore-forming protein (perforin) and an enzyme (granzyme B)
• The tumor cell enters death within 10–15 minutes
• The same NK cell can then move to a new target
Bottom line: A single NK cell can destroy 20+ tumor cells in sequence — fast and economical. Unlike many immune cells, it is not "single-shot."
Prager et al., J Exp Med, 2019; 216(9):2113-2127Click cards for details
NK cell therapy has one of the lowest adverse-effect profiles among cellular immunotherapies. Safety data validated in clinical studies.
No cytokine release syndrome (CRS) has been observed in patients receiving NK cell therapy in clinical trials. NK cells generate a controlled immune response.
No adverse effects on the central nervous system have been reported. NK cells do not cause off-target tissue damage.
No graft rejection occurs with allogeneic (donor-derived) use. NK cells can be safely administered from different donors.
Pre-manufactured "off-the-shelf" therapy is feasible. No patient-specific production required, enabling rapid treatment initiation.
By operating independently of MHC, NK cells are resistant to antigen-escape mechanisms. They recognize and destroy targets through multiple pathways.
A single NK cell can detach unharmed and eliminate up to ~20 target cells in succession. It remains active throughout the course of treatment.
Liu et al., NEJM 2020 • Myers & Miller, J Hematol Oncol 2021
Six core approaches developed in the laboratory to enhance the cancer-fighting potential of Natural Killer cells.

Specific recruitment and activation of NK cells into the tumor microenvironment. Targeted homing via CXCR3 and CX3CR1 receptors.
Cell Migration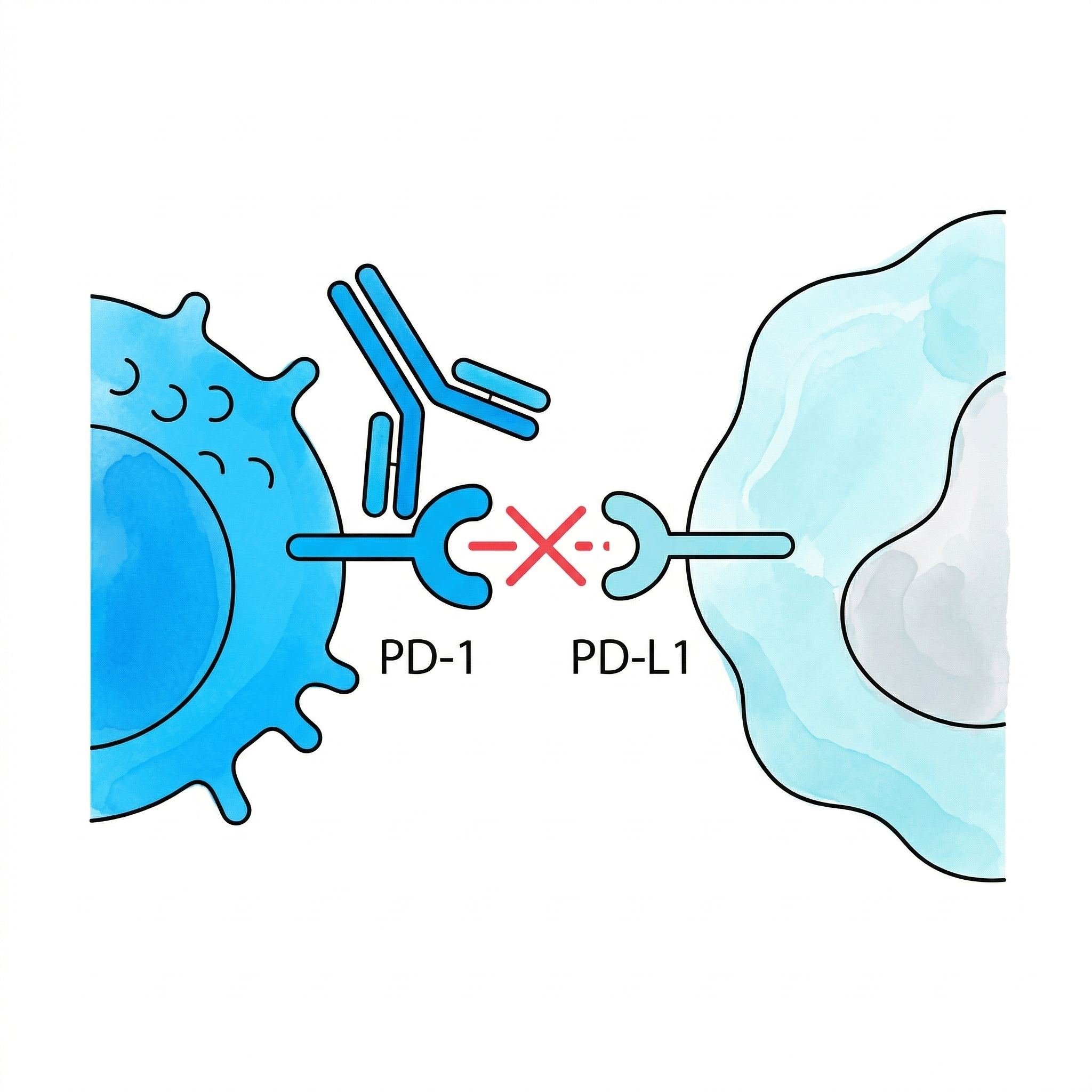
Blocking "brake" receptors such as PD-1 and CTLA-4. Anti-PD1 and anti-CTLA4 antibodies unleash NK cell activity.
Immune Escape Blockade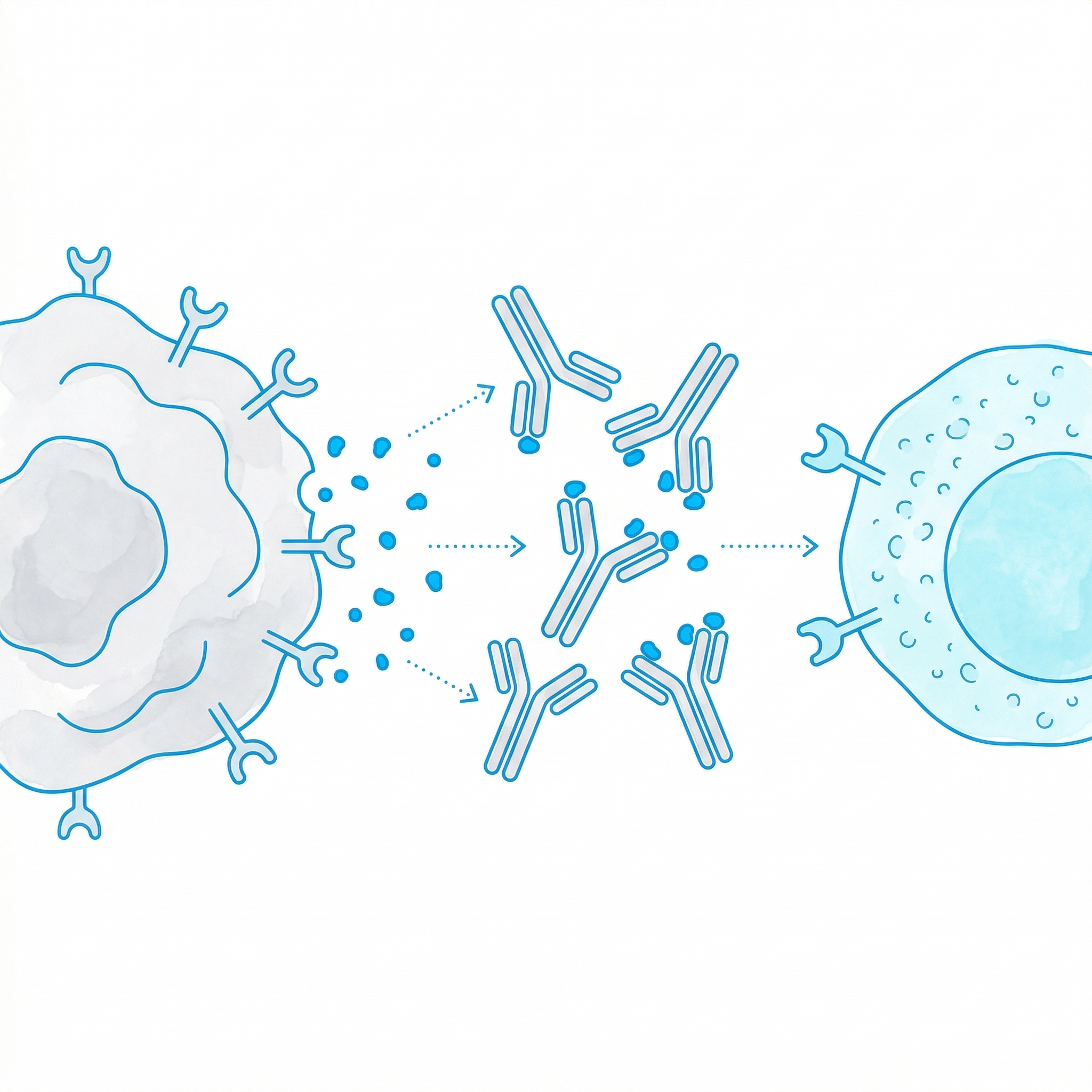
Antibody-mediated interventions that prevent tumor ligand shedding. Ligand-neutralizing antibodies disable the escape mechanism.
Ligand Blockade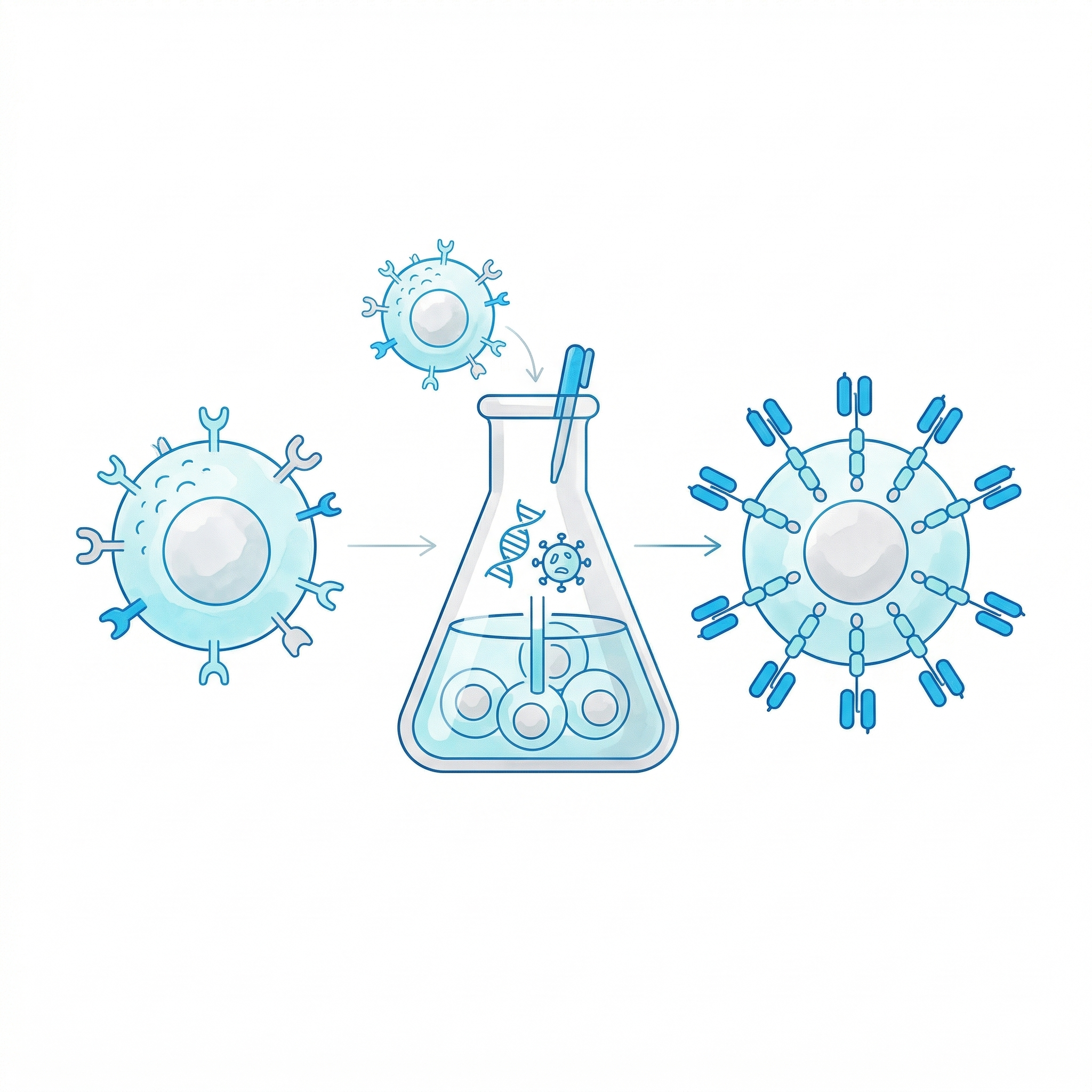
Transfer of genetically modified cells such as CAR-NK and CAR-T to the patient. Equipped with chimeric antigen receptors (CAR) for target-specific recognition.
Cell Engineering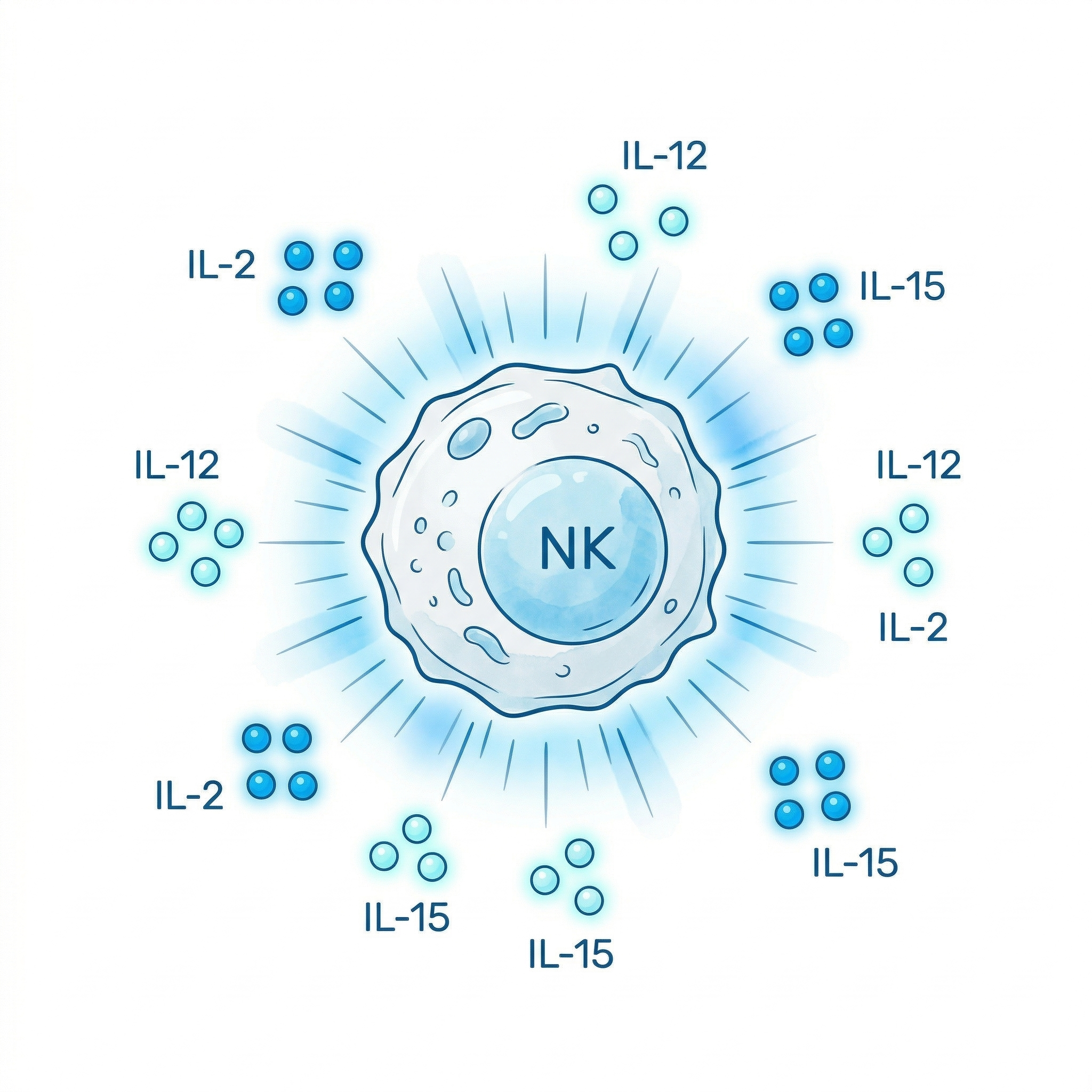
Cellular priming, expansion, and activation with IL-2, IL-12, and IL-15. CIK (Cytokine-Induced Killer) cells are generated ex vivo using this approach.
Cytokine Therapy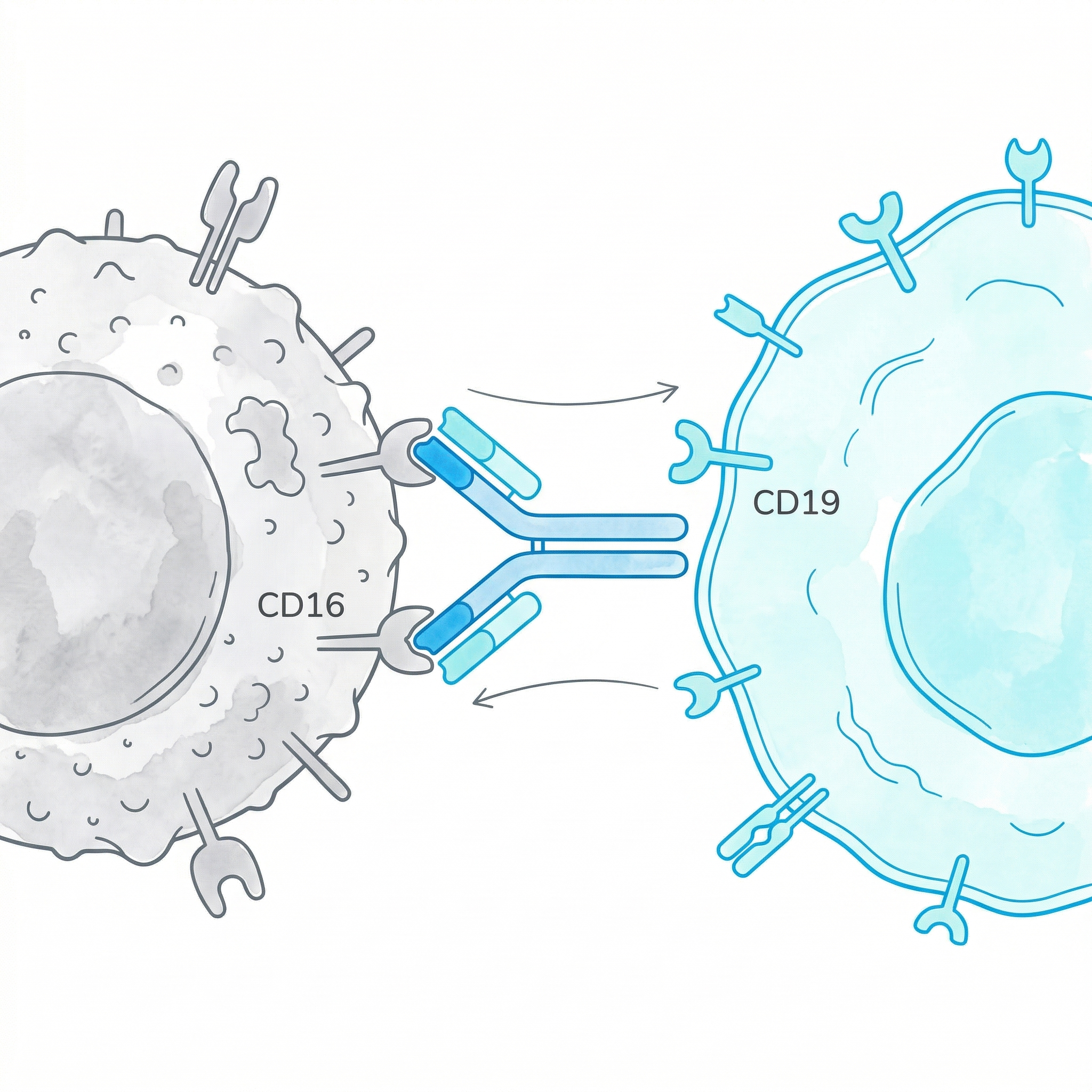
Specialized bridging molecules that directly link NK cells to cancer cells. Simultaneous capture via CD16 and CD19.
Targeted BindingWe develop and manufacture NK cells in our own R&D laboratory in compliance with international standards. Domestic production, global quality.

We develop and produce NK cells in our own GMP-compliant R&D facilities without reliance on external sources.
We manage every process under one roof, from cell isolation and expansion to quality control and clinical application.
GMP (Good Manufacturing Practice) compliant processes, international certifications, and continuous audit infrastructure.
We manufacture ministry-approved immunotherapy products in our own laboratory, including NK cells, CIK, and Dendritic Cells.
NK cells have a significantly safer profile compared to other cellular therapies such as CAR-T. The risks of cytokine release syndrome (CRS) and neurotoxicity, which are frequently observed with CAR-T therapy, are extremely low with NK cell treatment. This is because NK cells, as part of the innate immune system, generate a more controlled immune response.
NK cells are being investigated in clinical trials against more than 20 different cancer types. Particularly high response rates have been observed in hematological malignancies such as leukemia and lymphoma. Promising results are also emerging in solid tumors (lung, ovarian, colorectal).
CAR-T therapy requires genetic modification of the patient's own T cells -- a personalized, costly, and time-consuming process. NK cells, on the other hand, can be used allogeneically (from a donor), making pre-manufactured "off-the-shelf" therapy possible. This lowers cost, improves access, and shortens time to treatment initiation.
Genkord has been active in the field of cell biology since 2003. The company plans to produce NK cells in its own cGMP-certified laboratory, with end-to-end control from isolation to expansion. Currently, Genkord manufactures more than 15 biological products (mesenchymal stem cells, exosome series) under the Veragen brand.
NK cell therapies are currently being investigated in more than 500 clinical trials worldwide. Some NK-based treatments have reached Phase II and Phase III stages. At Genkord, we closely follow developments in this field and are preparing our manufacturing infrastructure accordingly.
Unlike T cells, NK cells do not carry a risk of GvHD (Graft-versus-Host Disease). This is one of the key features that makes allogeneic (different-donor) use of NK cells safe. Donor-derived NK cells do not attack the recipient's healthy tissues -- they only target abnormal cells.
Contact our team to learn more about NK cell therapies, R&D collaborations, or clinical trial partnerships.