CD3+CD56+ Effectors
Primary Effector
The main strike force of CIK. Exhibits MHC-unrestricted cytotoxicity via NKG2D and DNAM-1 receptors. Eliminates tumor cells through dual pathways (perforin/granzyme + Fas/FasL).
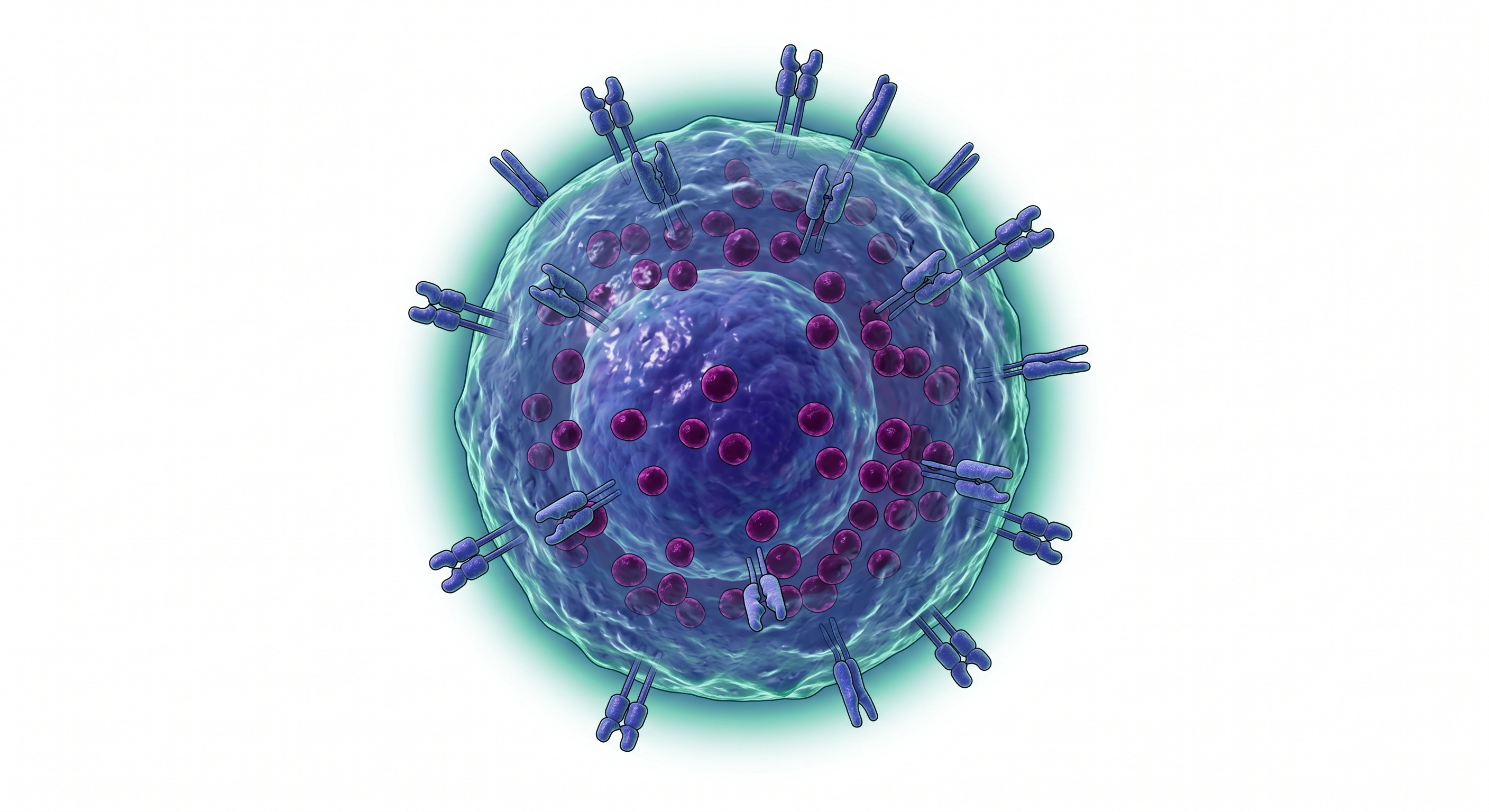
Hybrid immune effectors that combine the MHC-unrestricted target recognition of NK cells with the adaptive response capability of T cells.
Discover CIK Cells ↓CIK (Cytokine-Induced Killer) cells are a heterogeneous effector cell population generated by activating peripheral blood mononuclear cells ex vivo with IFN-γ, anti-CD3 antibody, and IL-2. Thanks to their CD3+CD56+ dual-positive phenotype, they carry the weapons of both innate and adaptive immunity within a single cell.
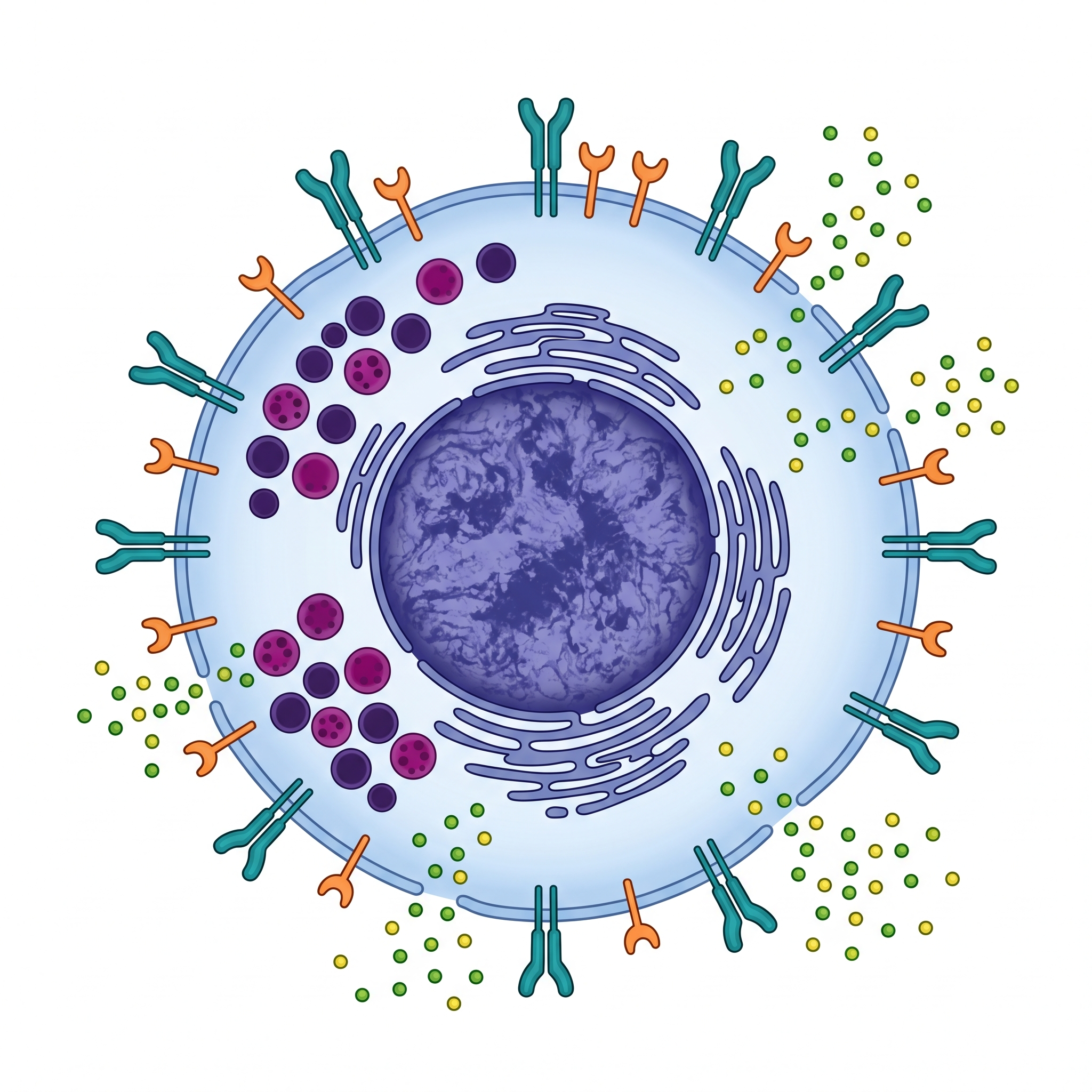
Click the numbered hotspots on the left to explore different regions of the CIK cell.
Hybrid Surface Markers
The primary effector population of CIK cells co-expresses both the T cell marker CD3 and the NK cell marker CD56. This dual-positive phenotype confers a unique "NKT-like" character, combining the advantages of both arms of immunity.
MHC-Unrestricted Recognition
The primary target recognition mechanism of CIK cells operates through the NKG2D receptor. This receptor recognizes MICA/MICB and ULBP ligands on stressed and malignant cells, triggering MHC-unrestricted cytotoxicity — neutralizing tumor immune evasion attempts that rely on downregulating antigen presentation.
Antigen-Specific Recognition
CIK cells carry a functional T cell receptor linked to the CD3 complex. This provides antigen-specific adaptive recognition in addition to NKG2D-mediated innate detection. The dual pathway offers a double safeguard against tumor escape mechanisms.
Perforin & Granzyme B
Like NK cells, CIK cells contain dense cytotoxic granules. Perforin forms pores in the target cell membrane while Granzyme B enters through these pores to trigger the caspase cascade and initiate apoptosis. CIK cells detach unharmed and can serially destroy multiple targets.
IFN-γ, TNF-α, IL-2
CIK cells are potent cytokine producers. The IFN-γ they secrete activates macrophages, TNF-α disrupts tumor vasculature and sensitizes the tumor microenvironment to immune attack. This "cytokine bridge" recruits adaptive immunity, orchestrating a systemic immune response.
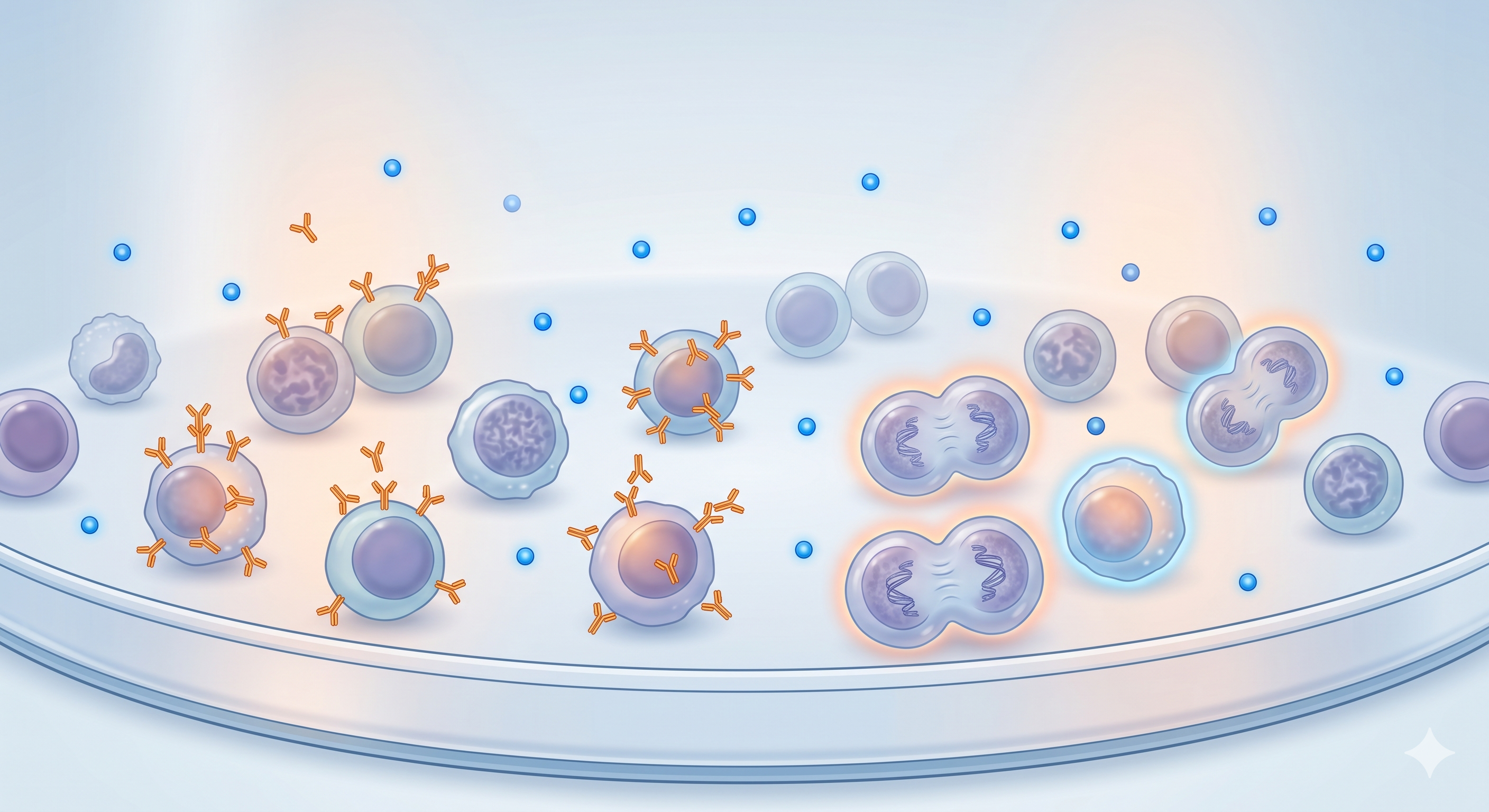
A CIK culture is not a single cell type — it is a coordinated ecosystem of three distinct immune cell populations. This heterogeneity is itself an advantage: a tumor cannot escape all three with a single evasion strategy.
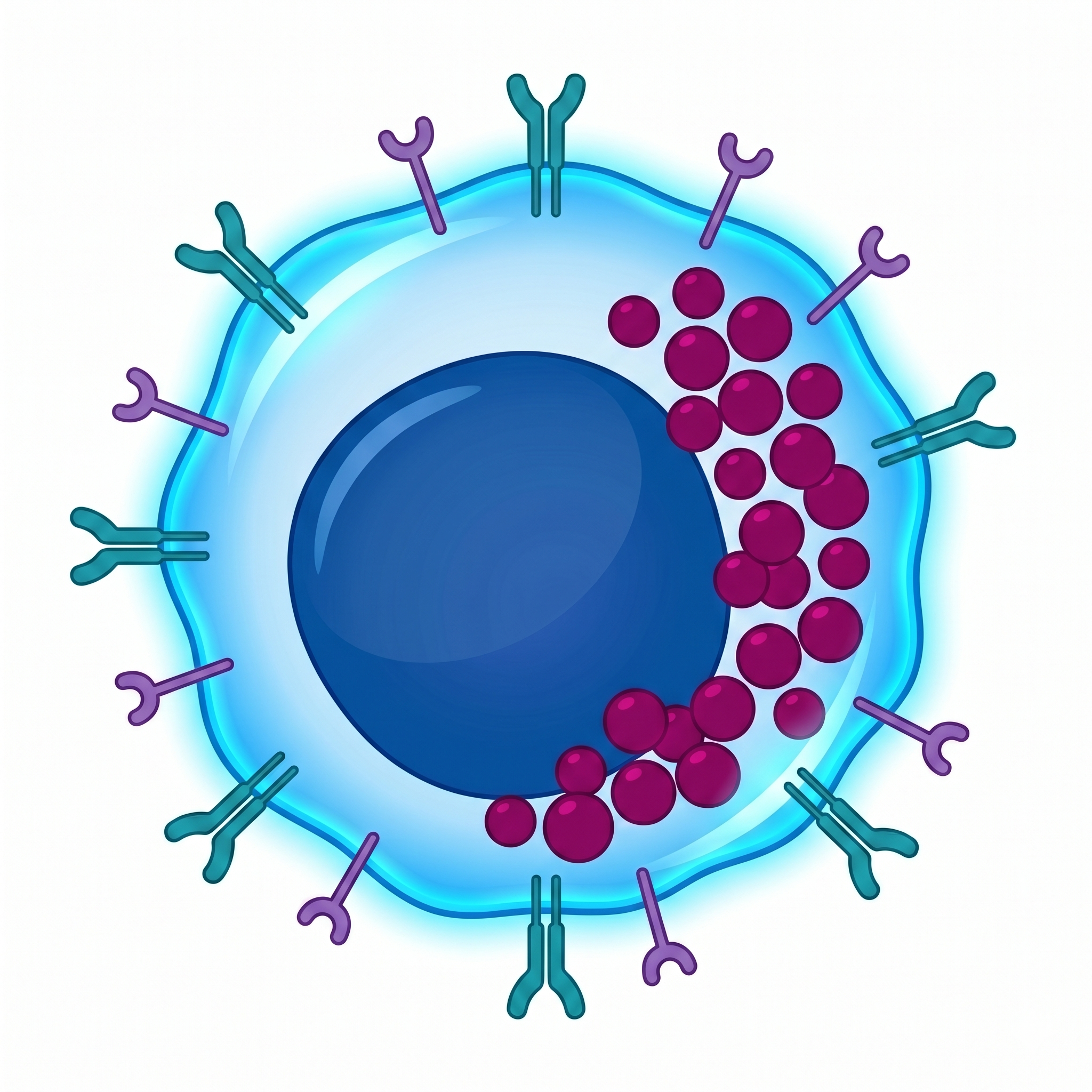 CD3⁺CD56⁺
20-40%
CD3⁺CD56⁺
20-40%
 CD3⁺CD56⁻
50-60%
CD3⁺CD56⁻
50-60%
 CD3⁻CD56⁺
5-10%
CD3⁻CD56⁺
5-10%
Primary Effector
The main strike force of CIK. Exhibits MHC-unrestricted cytotoxicity via NKG2D and DNAM-1 receptors. Eliminates tumor cells through dual pathways (perforin/granzyme + Fas/FasL).
Adaptive Support
The most abundant population in the culture. Provides long-term protection through TCR-mediated antigen-specific recognition and immunological memory formation.
Innate Reconnaissance
Conventional NK cells that provide rapid cytotoxicity upon first contact. Small in number but significant in impact.
One of the greatest strengths of CIK therapy is its remarkably simple and well-defined manufacturing process. No genetic modification is required — just the right cytokines and patience.
Culture Initiation
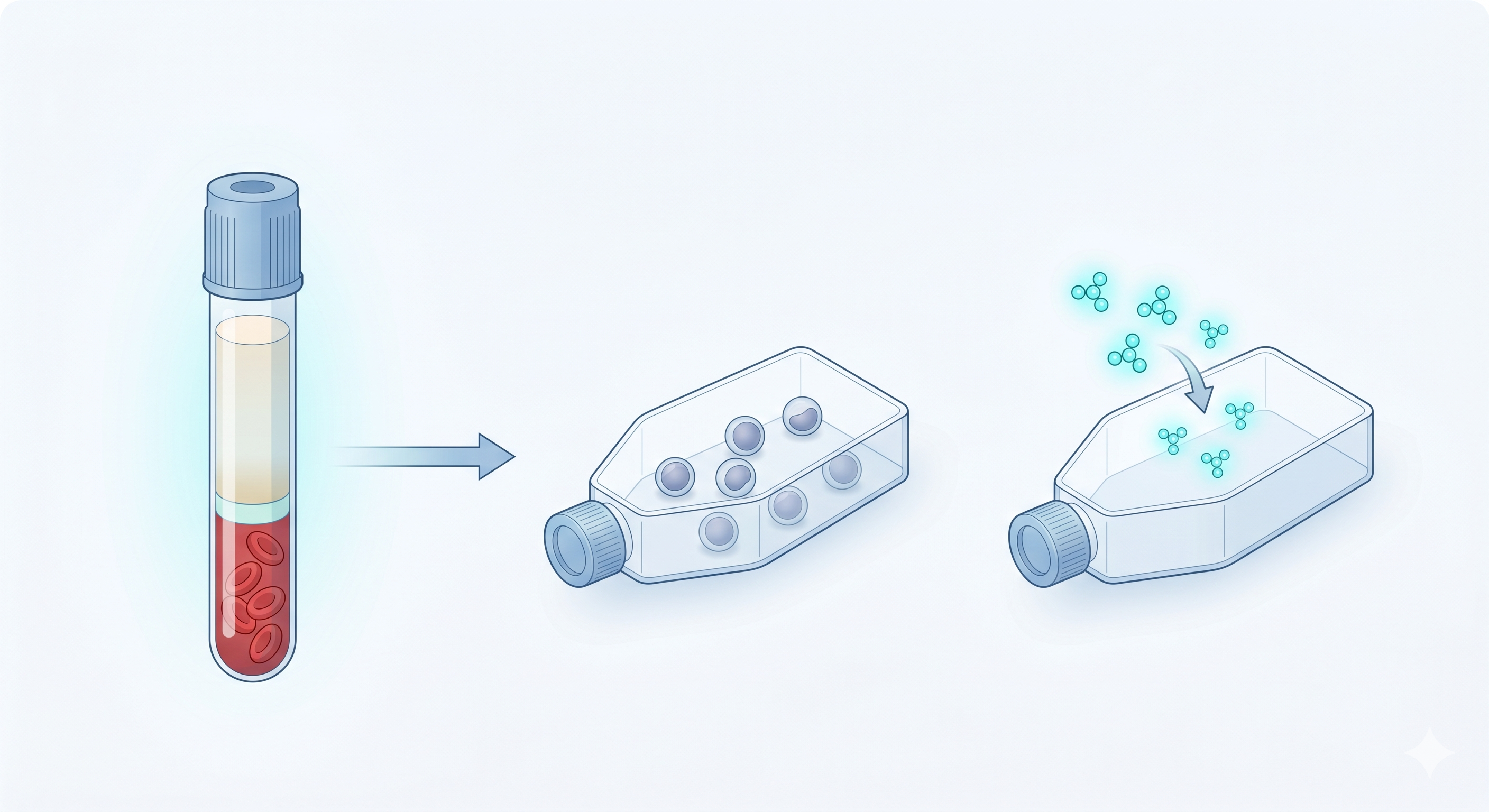
Mononuclear cells (PBMCs) are isolated from peripheral blood using Ficoll density gradient centrifugation and placed in serum-free culture medium (X-VIVO 15). In the first step, IFN-γ is added at 1000 U/mL. This priming phase prepares the cells for subsequent mitogenic stimulation and enhances IL-2 response capacity — the 24-hour pre-treatment is critically important.
Cellular Transformation
On the second day, two critical components are added: Anti-CD3 monoclonal antibody (OKT-3, 50 ng/mL) activates T cells, while IL-2 (300-600 U/mL) triggers proliferation. This combination initiates the transformation of PBMCs into the CIK phenotype. Cells begin to proliferate and acquire CD3+CD56+ surface expression.
Harvest and Release
Over two to three weeks, cells are expanded by adding IL-2 and fresh media every 2-3 days. By the end of this period, approximately a 1,000-fold expansion from the starting count is achieved. The CD3+CD56+ fraction rises to 20-40%. Prior to harvest, flow cytometry phenotype analysis, sterility testing, endotoxin measurement, and viability assessment are performed.

Clinically proven outcomes tested on more than 10,000 patients worldwide.
230 patients with liver cancer (hepatocellular carcinoma, HCC) cleared by surgery or thermal ablation were randomized: half received standard follow-up only; the other half received standard follow-up plus CIK cell therapy:
• In the CIK group, cancer recurrence risk was 38% lower (Hazard Ratio 0.63; 95% confidence interval: 0.43-0.94; p=0.010 — statistically robust)
• Recurrence-free survival: 44 months with CIK vs 30 months in controls
Bottom line: When CIK is given after surgery/ablation, it delays cancer recurrence by an average of 14 months. A promising adjunct to standard care in liver cancer.
Lee JH, et al., Gastroenterology, 2015; 148:1383-1391The International Registry on CIK Cells (IRCC) is the official database aggregating worldwide clinical data on CIK therapy. 10-year survey:
• 106 clinical studies registered
• 10,225 patients tracked
• 4,889 patients treated directly with CIK cells
• 30+ different cancer types targeted
Bottom line: CIK is not a small experimental method. With an evidence base spanning tens of thousands of patients, it is a standardized cellular therapy used at multiple international centers.
Zhang Y, Schmidt-Wolf IGH, J Cell Physiol, 2020CIK cells have been tested across 30+ different cancer types. Strongest clinical evidence in:
• Liver cancer (hepatocellular carcinoma, HCC)
• Colorectal cancer
• Kidney cancer (renal cell carcinoma)
• Lung cancer (NSCLC)
Meta-analysis pooled from 70 studies and 6,743 colorectal cancer patients: with CIK, overall survival Hazard Ratio 0.59, disease-free survival HR 0.55.
Bottom line: Because CIK combines features of two immune cell types (NK + T cell), adding it to standard care reduces mortality risk by 41%. Demonstrates applicability across many cancer types.
Li CMY, et al., J Immunother Cancer, 2023CIK manufacturing works as follows: immune cells from the patient's blood are stimulated in the lab with specific cytokines (IL-2, IFN-γ, anti-CD3 antibody):
• Total production time: 14-21 days
• During this time cells expand hundreds to 1,000-fold
• No genetic engineering required (unlike CAR-T)
Bottom line: CIK can be prepared in about 3 weeks under international pharmaceutical manufacturing standards (cGMP). Far simpler and more affordable than CAR-T — accelerating access to therapy.
Sharma P, Schmidt-Wolf IGH, J Exp Clin Cancer Res, 2021; 40:341Click the cards for details
Over 20 years of clinical experience have demonstrated the superior safety profile of CIK cell therapy. Meta-analyses confirm that CIK therapy does not increase serious adverse events compared to standard treatment.
Severe cytokine release syndrome (CRS), commonly seen with CAR-T therapy, is extremely rare and mild with CIK cells. Produces a controlled immune response.
No adverse effects on the central nervous system have been reported. CIK cells do not cause off-target neurological damage.
CIK cells preserve healthy tissues through NKG2D-mediated target recognition. GvHD rates remain low even in allogeneic settings.
No genetic modification or viral vectors required. Manufactured in 14-21 days under standard GMP conditions using a cytokine cocktail. Cost-effective compared to CAR-T.
Thanks to the broad ligand recognition capacity of the NKG2D receptor, CIK cells exhibit anti-tumor activity against more than 30 different cancer types.
CIK cells attack through two distinct pathways: granule release (perforin) and death signaling (Fas/FasL). If one pathway is blocked, the other takes over.
Lee et al., Gastroenterology 2015 • Zhang & Schmidt-Wolf, J Cell Physiol 2020 • Sharma & Schmidt-Wolf, J Exp Clin Cancer Res 2021
Dendritic cells (DCs) process tumor antigens and present them to CIK cells. This bidirectional interaction generates a potent anti-tumor response that neither modality can achieve alone.
Collects tumor-associated antigens, processes them, and prepares them for presentation via MHC molecules.
DCs present processed tumor antigens to CIK cells. The bidirectional interaction enhances IL-12 secretion, boosting cytotoxicity.
Armed with antigen information, CIK cells mount enhanced targeted cytotoxicity through both NKG2D and TCR-mediated dual pathways.
Lin et al., Immunol Lett 2017 • Jiang et al., J Transl Med 2024
Genkord is one of the rare facilities that manufactures both DC and CIK cells in its own cGMP-certified laboratory.
We develop and manufacture CIK cells in our own R&D laboratory to international standards. Local production, global quality.

We develop CIK cells in our own GMP-compliant R&D facilities, without reliance on external sources.
From blood collection to CIK harvest, from quality control to clinical application — all processes managed under one roof.
GMP-compliant processes, international certifications, and continuous audit infrastructure.
One of the rare facilities capable of manufacturing three distinct cellular therapy platforms — NK, CIK, and Dendritic Cell — in the same laboratory.
CIK (Cytokine-Induced Killer) cells are produced by activating mononuclear cells isolated from the patient's or a healthy donor's peripheral blood with IFN-γ, anti-CD3 antibody, and IL-2 cytokines in the laboratory. Over a 14-21 day culture period, cells expand approximately 1,000-fold and acquire potent anti-tumor activity. Since no genetic modification is required, the manufacturing process is far simpler and more cost-effective than CAR-T therapy.
NK cells rely exclusively on innate immune mechanisms, whereas CIK cells combine both NK and T cell properties. The primary effector population of CIK — CD3+CD56+ cells — performs MHC-unrestricted recognition via the NKG2D receptor (NK-like), while also capable of antigen-specific responses through the TCR (T-like). This dual mechanism provides an advantage against tumors resistant to a single pathway.
Yes. Over 20 years of clinical experience have demonstrated the excellent safety profile of CIK cell therapy. The most common side effects are mild fever (Grade 1-2) and transient fatigue. The risks of severe cytokine release syndrome (CRS), neurotoxicity, and severe GvHD — frequently associated with CAR-T therapy — are extremely low with CIK treatment. Meta-analyses indicate that the rate of serious adverse events is below 5%.
CIK cell therapy has been investigated in more than 30 different cancer types. The strongest evidence base exists for hepatocellular carcinoma (HCC), colorectal cancer, renal cell carcinoma, and NSCLC. Randomized controlled trials have shown that CIK therapy significantly improves recurrence-free survival, particularly when used as adjuvant therapy following surgery or chemotherapy.
DC-CIK is a combination therapy in which Dendritic Cells (DCs) and CIK cells are used together. Dendritic cells process tumor antigens and present them to CIK cells — this antigen presentation enables CIK cells to mount a more targeted and potent cytotoxic response. Clinical studies have demonstrated that the DC-CIK combination achieves higher response rates than CIK therapy alone. Genkord manufactures both DC and CIK in the same laboratory.
CAR-T therapy requires viral vector-mediated genetic modification, a patient-specific manufacturing process, and weeks of complex protocols — in the US, the cost per treatment can exceed $300,000. CIK cells require no genetic modification: they are produced in a GMP setting using only clinical-grade cytokines (IFN-γ, IL-2, anti-CD3) within 14-21 days. This standardized and straightforward protocol significantly reduces manufacturing costs and improves accessibility.
Contact our team to discuss CIK cell therapies, DC-CIK combinations, R&D collaborations, or clinical study partnerships.